China3D printingNet September 29th, the leading3D printingMachine OEM 3D Systems’ VSP orthopedics platform has obtained 510(k) approval from the U.S. Food and Drug Administration (FDA). VSP Orthopedics is exclusively provided through Onkos Surgical’s My3D™ personalized solution platform for virtual surgical planning of complex procedures.It can be combined with other professional fields of 3D Systems to design and3D printingAnatomical models and cutting guides to provide further surgical assistance.
The platform is part of 3D Systems’ strategic approach, aiming to establish a strong position in the 3D healthcare field.Recently, the company also obtained a 510(k) license for its D2P™ (DICOM-to-PRINT) software, which is specifically designed to help clinicians3D printingAnatomical model.
Radhika Krishnan, senior vice president of advanced software and healthcare at 3D Systems, said: “Based on our success in using personalized healthcare solutions in other surgical specialties, we are confident that this technology will have a positive impact on the orthopedics community.”
3D Systems Virtual Surgery Planning
VSP Orthopedics is part of a broader virtual surgery planning solution provided by 3D Systems. According to reports, the foundation of 3D Systems VSP was laid 25 years ago, and since then, more than 120,000 patients have been helped. As of January 2018, Stryker, an American medical technology company, has partnered with 3D Systems to provide a series of 3D Systems virtual surgery planning options with the assistance of professional biomedical engineers. However, at present, VSP orthopedic products can only be obtained through the technology provided by Onkos Surgical.
After obtaining a 510(k) clearance from the FDA, it now means that VSP Orthopedics is “at least as safe and effective” for another legally marketed device, indicating that it is suitable for safe applications.
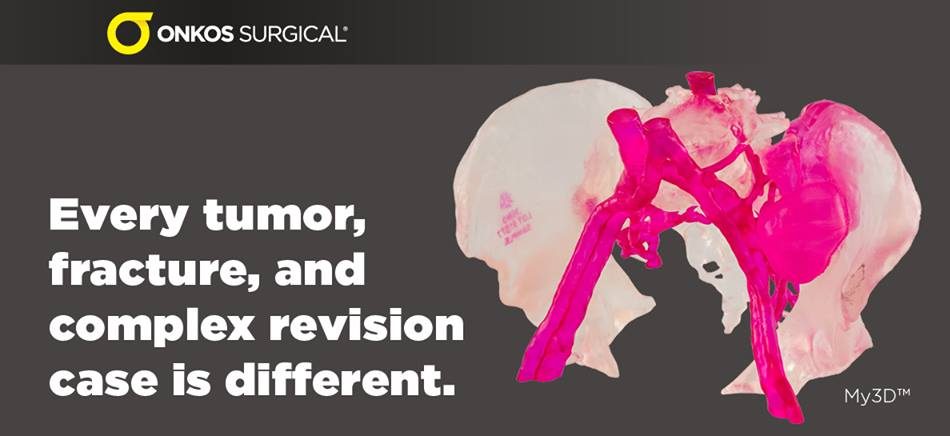
Onkos Surgical My3D™ personalized solution platform. Picture from Onkos Surgical
Improve surgical results
Founded in 2015, Onkos Surgical is a professional provider of medical implants and devices for the treatment of patients with musculoskeletal tumors. The company is headquartered in Parsippany, New Jersey. In 2017, it received $17.6 million in Series B financing from three investors including 3D Systems.
Onkos’ My3D™ personalized solution platform is used to develop personalized implant, instrument and anatomical model designs for surgeons. With the help of the integrated VSP orthopedics, the platform increases the ability of precise tumor resection planning, and in some cases can preserve natural joints. This solution can help improve surgical results and save operating room time. Patrick Treacy, CEO of Onkos Surgical, said,
“As an early investor in Onkos, 3D Systems understands our passion for development and innovation.
The VSP orthopedic system can only be used through our My3D personalized solutions platform, which is a good example of how we can use advanced surgical planning and personalized features to create novel solutions that musculoskeletal patients deserve. “

3D Systems’ FDA-cleared VSP Orthopedics solution enables surgeons to obtain clear 3D visualizations of the patient’s anatomy and develop a personalized surgical plan before entering the operating room.
In addition to My3D™, Onkos also has uDesign™ planning software for integrated implants, ELEOS™ limb rescue system implants, MyTray™ instrument manager and GenVie™ biological products.
China3D printingNet original article!
(Editor in charge: admin)


0 Comments for “3D Systems virtual surgery planning platform is licensed by the U.S. Food and Drug Administration”